Bivalent vaccine
BA4 and BA5 share many similarities to the original variant but also appear to have mutations that make them. In this ongoing phase 2-3 study we.

Moderna Says Its New Bivalent Vaccine Shows Promise Shots Health News Npr
Original COVID-19 vaccines which have been authorized and administered to millions of Americans since 2020 are now referred to as monovalent According to the FDA.
. A bivalent vaccine elicits an immune response against two different antigens. Isaac Bogoch expects we could be running three simultaneous vaccine programs. A booster program a.
In Study 4 a total of 610 participants greater than 55 years of age previously vaccinated. Kaiser nurse Marilyn Antonio left gives a Moderna booster shot to Ted Naifeh. If bivalent shots are ready for fall infectious disease expert Dr.
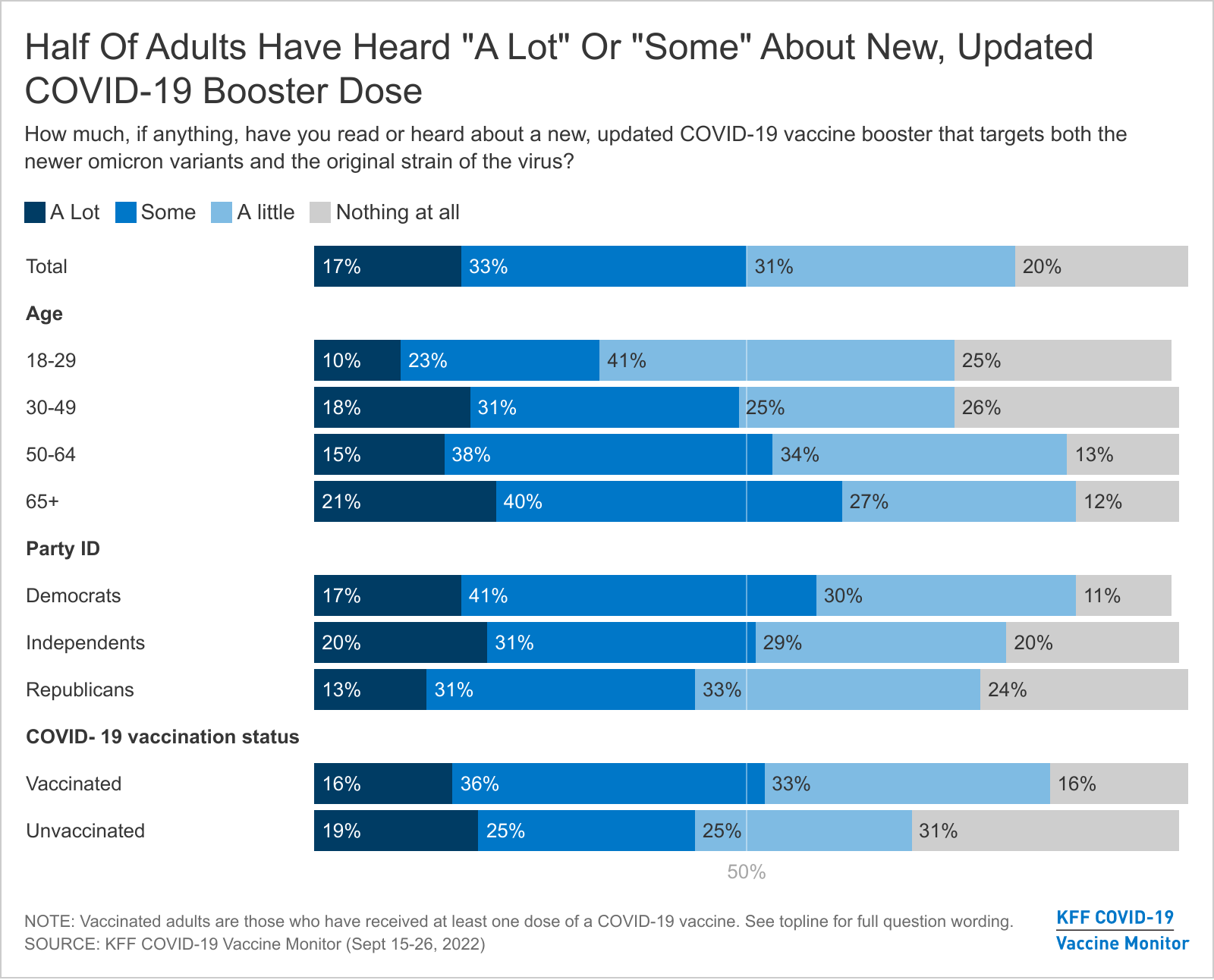
As of Sept. About 230 million Americans are eligible for a bivalent COVID-19 vaccine booster. The new bivalent COVID-19 vaccine includes mRNA from the original strain of SARS-CoV.
The bivalent vaccine which Moderna has said it hopes will be authorized for use in the United States this fall is designed to target both the original omicron variant and the. 1 the Centers for Disease Control and Prevention authorized an updated COVID-19 vaccine with the goal of fending off a surge in cases this fall and winter. Bivalent Vaccine Original and Omicron BA1 Administered as a Second Booster Dose.
The safety and immunogenicity of the bivalent omicron-containing mRNA-1273214 booster vaccine are not known. The FDA authorized bivalent formulations of the Moderna and Pfizer-BioNTech COVID-19 vaccines for use as a single booster dose at least two months after completing. Bivalent Covid-19 booster jabs extended to those aged 18 to 49 from Nov 7.
Based on post market surveillance to date over 11 million updated bivalent mRNA vaccine doses have been administered in the US without any safety concerns reported. New bivalent COVID vaccine may not be more protective but its still recommended. Health Canada has approved Pfizers new bivalent COVID-19 vaccine which targets the virus strains now most common in Canada.
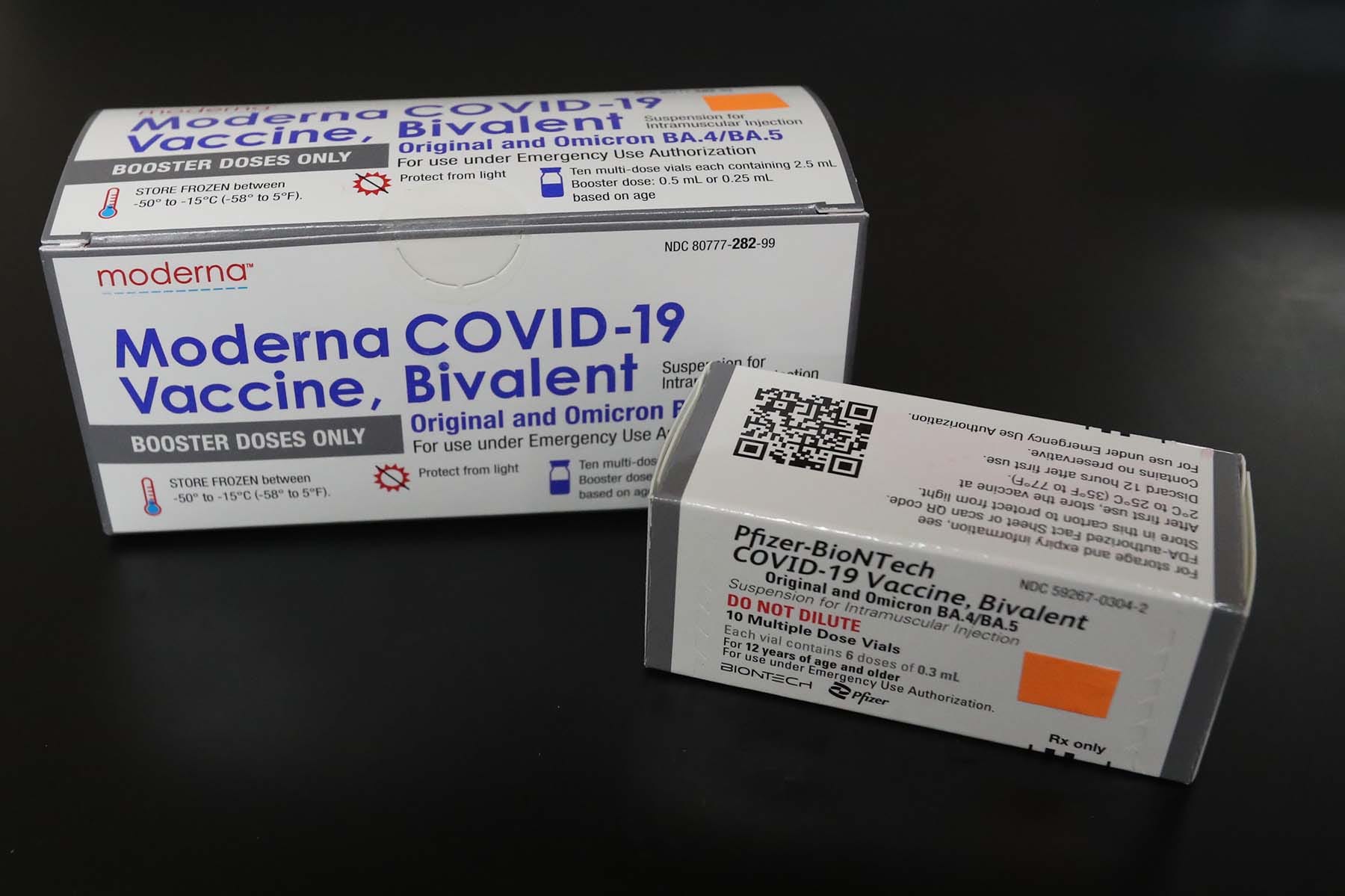
If you recall on August 31 the FDA announced that they had amended the original Moderna and Pfizer-BioNTech Covid-19 mRNA vaccine EUAs to include their new bivalent. The updated version of the Pfizer-BioNTech. It contains both the original vaccine strain of the virus and a strain derived from the BA5.
A bivalent vaccine however contains mRNA components from two strains of virus. The bivalent booster is the most recent version of the COVID-19 vaccine. Pfizers bivalent booster is authorized for those 5 or older while Modernas authorization is for.
This can means two different viruses or two variations of one virus. The current COVID-19 vaccines. Data collected by the FDA for earlier bivalent COVID-19 booster vaccines suggests that these shots successfully provided immunogenicity a boost to your immunity and elicited.
The bivalent vaccine targets the original omicron variant. To evaluate the overall effectiveness of bivalent booster vaccines a clinical study used a bivalent COVID-19 vaccine that targeted both the original strain of COVID-19 as well as a different. They compared virus neutralization by sera collected from individuals vaccinated with three doses of the original monovalent vaccines and a fourth dose of the bivalent vaccine.
The safety of a single booster dose of the Pfizer-BioNTech COVID-19 Vaccine Bivalent for individuals 12 years of age and older is based on safety data from a clinical study. An updated version of the COVID-19 vaccine made by Moderna that targets two coronavirus variants known as a bivalent vaccine has today been. Households to get antigen rapid test kits.
New Bivalent Covid Vaccine Booster
Covid 19 Bivalent Vaccine Available At Uhc Uga Student Affairs
What You Need To Know About Bivalent Boosters For Covid 19 Hub
Updated Covid 19 Bivalent Booster Shots Now Available In Coachella Valley
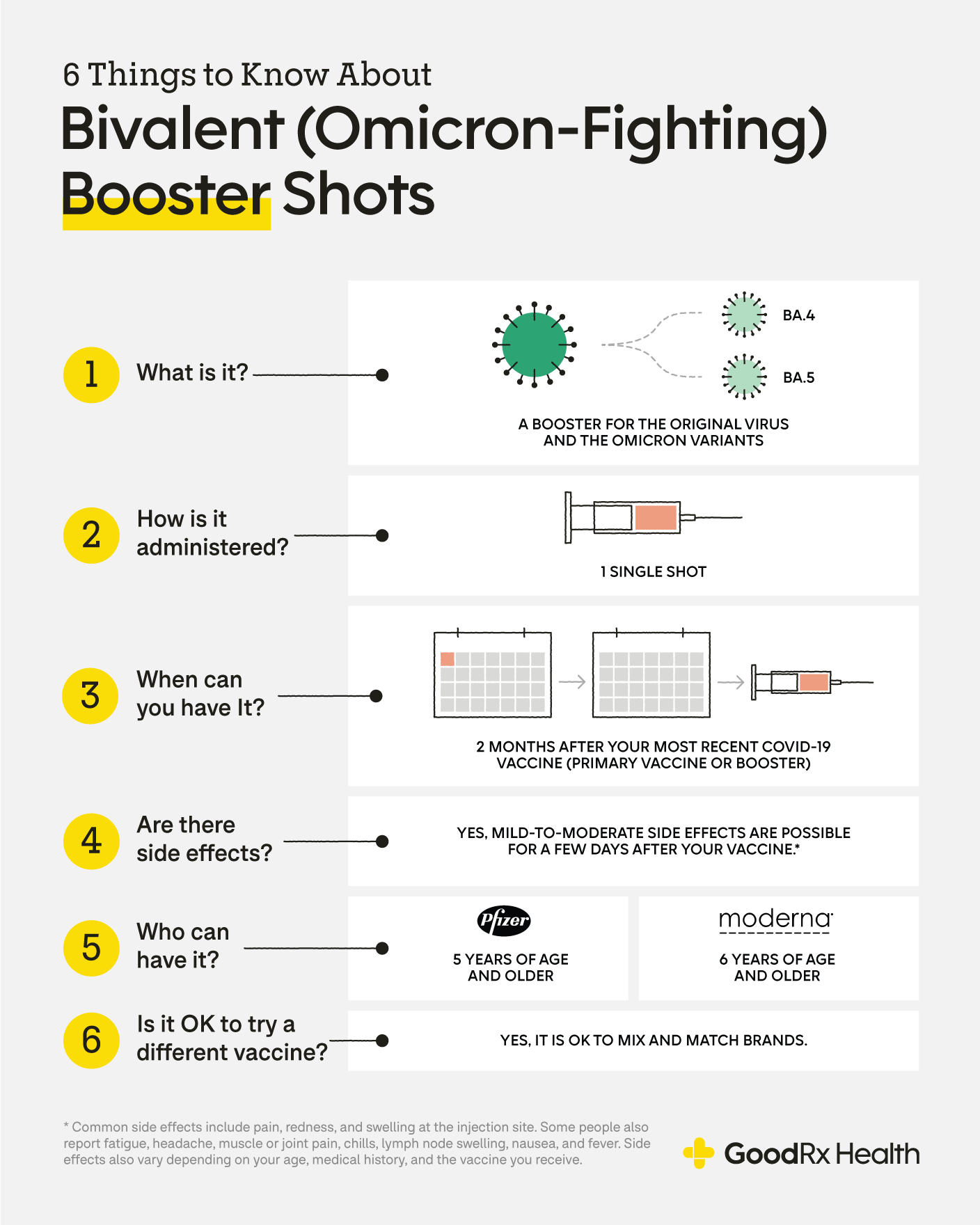
What S The Most Effective Covid 19 Booster Shot Available Today Goodrx
Covid 19 Bivalent Booster In Michigan Where To Get It
Vaccines Boosters Fishers In Official Website

Moderna S Bivalent Booster Becomes First Authorized Omicron Vaccine

Covid 19 Vaccine Hawai I Doh Info Resources For Managing Covid 19
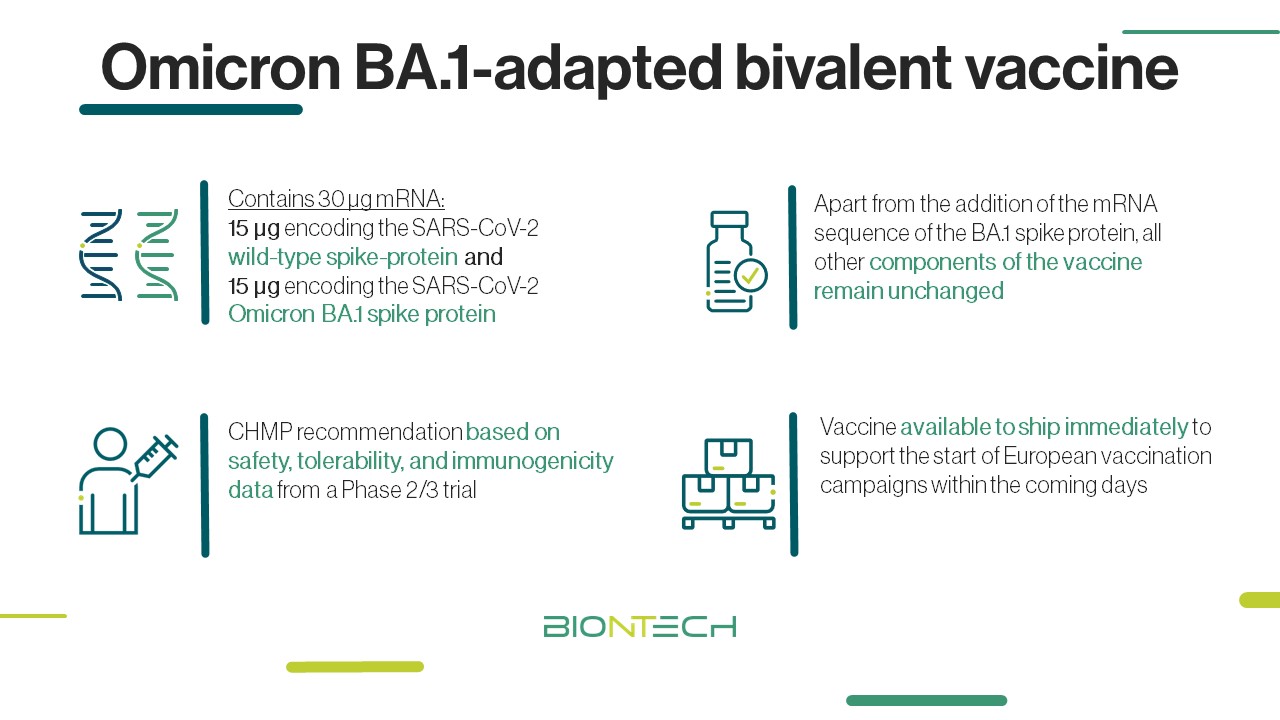
Biontech Se On Twitter Omicron Ba 1 Adapted Bivalent Vaccine Update Our Ba 1 Bivalent Covid 19 Vaccine Was Recommended For Conditional Marketing Authorization By Ema News Committee For Medicinal Products For Human Use As A Booster

New Covid Boosters What To Know About The Bivalent Vaccines That Offer Better Protection Against Omicron Colorado Public Radio
Pfizer Biontech Releases Early Results From Bivalent Booster Vaccine Trial
A Bivalent Recombinant Vaccine A Promising Strategy Against Both Sars Cov 2 Variants And Wild Type Of The Virus Signal Transduction And Targeted Therapy
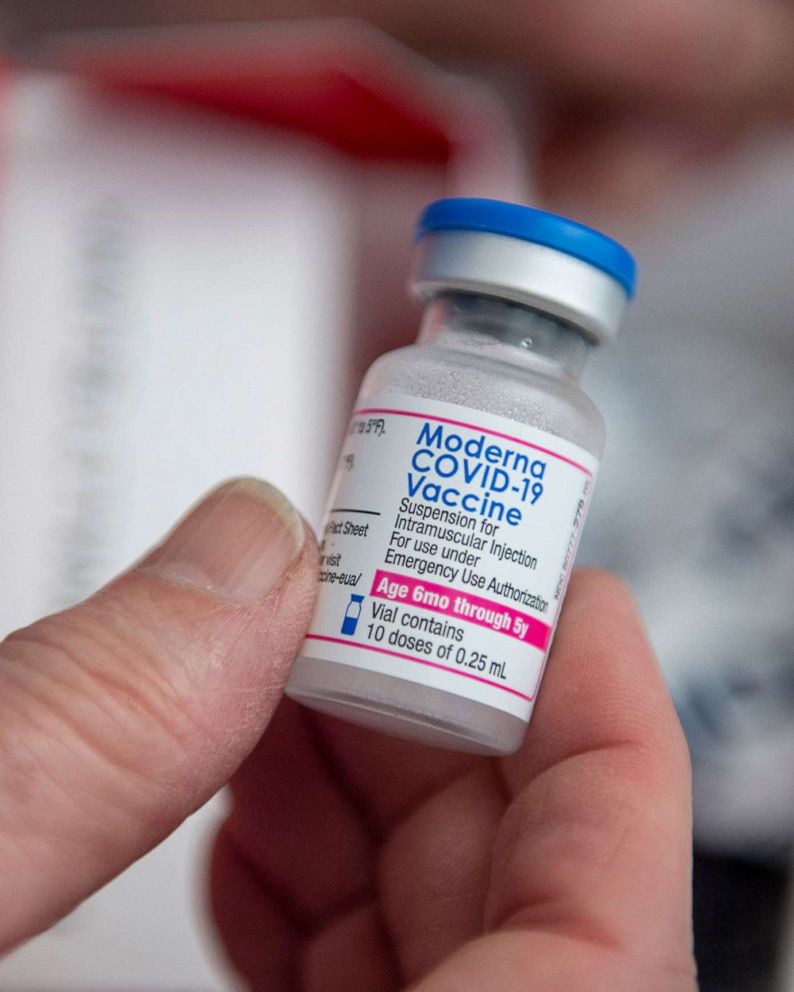
New Covid Bivalent Vaccine Expected In Us In The Fall Abc News
/cloudfront-us-east-1.images.arcpublishing.com/gray/CKOLGG4UVNDSPN2RHYCDVTCNOI.jpg)
Bivalent Covid Vaccine Could Be Available Soon Variants Continue To Spread

Ottawa County Covid 19 Update September 13 2022

Update On Covid 19 Vaccines And Evusheld Penn Medicine

Moderna Says Its New Vaccine Booster Shows Superior Response To Omicron Npr
